
March 10, 2026
As GLP-1 indications expand, accurate risk assessment depends on confirming diagnosis, companion therapy signals, and persistence in Prescription Data.
Underwriters ask a lot of questions about GLP‑1s, but no matter how they word them, they all want to know the same thing: What does this prescription actually tell me about risk?
A few years ago, that question usually had an obvious answer. Certain GLP‑1s almost certainly indicated diabetes. Others pointed to obesity. Then came a slew of new indications and off-label uses.
Today, GLP-1s are prescribed for an increasingly wide range of conditions. As a result, they are now one of the least self-explanatory drug classes underwriters see on a life insurance applicant profile. Interpreting them correctly requires context because the same prescription can reflect impairments with widely varying mortality.
GLP-1s are a “new” drug class that isn’t new at all
GLP‑1 medications may feel like an overnight success, but their story began decades ago. The first GLP‑1 drug was approved in 2005, after researchers discovered that compounds derived from Gila monster saliva could stimulate insulin release. That had obvious implications for the treatment of diabetes.
Then something unexpected happened. Patients taking GLP-1s weren’t just improving glycemic control; they were losing weight, and lots of it. That observation ultimately reshaped the trajectory of the entire drug class, leading to obesity-specific formulations and, more recently, FDA approvals for additional chronic conditions.
Since 2020, GLP-1 incidence in prescription histories has increased more than fivefold. Today, more than 11% of Irix Prescription Data hits show at least one GLP-1. From a clinical standpoint, that growth still has room to run. Underwriters are going to be seeing it even more often.
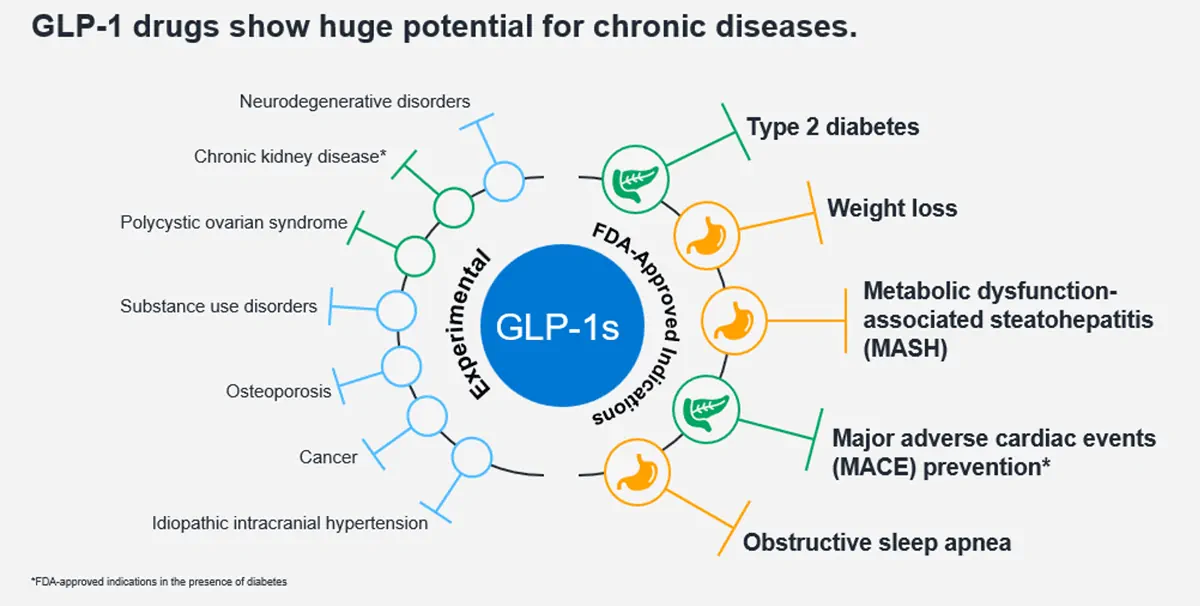
Several recently approved indications make it more important than ever to understand the underlying diagnosis. And with so much promising research underway, we’ve only just begun to identify conditions for which GLP-1s will be prescribed in the future.
GLP-1s no longer offer a risk-assessment shortcut
GLP‑1s are clinically powerful drugs that act on receptors throughout the body, including the pancreas, stomach, brain, cardiovascular system, and kidneys.
That wide-ranging effect is precisely why they’re now prescribed for such a broad set of conditions.
A GLP‑1 prescription could indicate:
- Well-controlled type 2 diabetes
- Severe obesity under active treatment
- Metabolic dysfunction associated steatohepatitis (MASH)
- Obstructive sleep apnea
- Elevated cardiovascular risk
- Chronic kidney disease
Clinically, those are very different scenarios with very different mortality implications. Treating the medication itself as the signal—without understanding why it’s prescribed—is where misinterpretation creeps in.
New indications underwriters can’t ignore
- Fatty liver disease (MASH)
Formerly known as NASH, metabolic dysfunction associated steatohepatitis is a form of fatty liver disease associated with significantly increased risks of diabetes, heart failure, kidney disease, cirrhosis, and liver cancer.
GLP-1s have rapidly become first-line therapy for many patients because they directly address the underlying driver: excess fat deposition in the liver. With FDA approval now in place, Wegovy is likely to appear more frequently in applicants with this diagnosis—sometimes as the medication signaling liver disease.
- Obstructive sleep apnea
In late 2024, Zepbound received FDA approval for obstructive sleep apnea. While GLP-1s don’t replace CPAP or BiPAP therapy, they meaningfully reduce apnea severity by driving weight loss.
For underwriters, the key is context: GLP-1s should appear alongside those respiratory machines referenced above. If they don’t, that’s a signal to dig deeper into diagnostic data.
- Cardiovascular and kidney outcomes
In patients with diabetes, GLP-1s are now FDA-approved to reduce major adverse cardiac events (MACE) and slow chronic kidney disease progression. Studies show:
- ~13% reduction in cardiovascular death
- Lower rates of heart attack and stroke
- Meaningful improvements in kidney outcomes
Initial evidence suggests that these benefits extend beyond diabetic populations, further broadening the underwriting implications.
Persistence: the clinical factor that changes the picture most
For underwriting, adherence matters as much as indication.
Up to half of all patients discontinue GLP‑1 therapy within a year, often due to side effects or cost. When treatment stops, benefits fade—and weight frequently returns.
That means two factors are critical:
2. Consistency – Are fills regular and persistent?
In some cases—particularly diabetes—long-term, consistent GLP-1 use may actually represent a positive underwriting factor compared to unstable or escalating therapy.
This isn’t a temporary shift
Even with current adoption levels, the market is far from saturated. A majority of individuals with severe obesity are not yet on GLP‑1 therapy. New oral formulations, expanding coverage, and additional FDA approvals are likely to push utilization higher.
In other words: this isn’t a peak, it’s a new baseline.
What this means for underwriting
GLP‑1s demand a shift in mindset. They can no longer be underwritten as proxy diagnoses.
When you see one, you must:
- Clarify the underlying condition using Irix Medical Data, Irix EHR, or by ordering an APS
- Assess how effective the drug is for that specific diagnosis
- Evaluate adherence and duration before assigning risk
Handled thoughtfully, GLP‑1 data can sharpen underwriting decisions rather than complicate them, but only if you know what you’re really looking at.
When those questions are answered, GLP‑1 data becomes an asset. Without that context, it’s easy to over‑ or under‑estimate risk.
Watch our webinar, Emerging clinical applications for GLP-1 receptor agonists, to learn more.
Key tips for underwriting applicants with GLP‑1 use
Immediately look for:
- Evidence of the underlying diagnosis (as seen in medical claims, EHR, APS)
- Whether the GLP-1 is FDA-approved or off-label for that condition
- Signs of medication adherence (≥12 months, regular fills)
Get cautious when:
- A GLP‑1 appears without any supporting clinical context
- Fills are intermittent or recently initiated
- There’s no alignment between the medication and expected companion therapies (e.g., no CPAP with sleep apnea)
Be confident when:
- The diagnosis is clear
- The drug is first line and clinically appropriate
- Use has been stable long enough to confer benefit
Kyle Schimek, PharmD
Kyle Schimek is a board-certified pharmacist and Clinical Services Manager at Milliman IntelliScript. He works closely with underwriting teams to translate prescription and medical claims data into clinically meaningful insight, helping insurers interpret emerging drug trends—like GLP-1s—within the context of real-world risk.



